 
PRACTICE PROBLEMS : Use the electron dot structure (Lewis structure) and the electron geometry table to determine the steric number and electron geometry. What is the steric number and electron geometry of PH5? What is the steric number and electron geometry of BrF3? Unformatted text preview: Molecular Geometry Chart of Electron Number of Electron Pair Groups Molecular Geometry Lone Pairs Approximate Arrangement 2 Bond Angles linear 180 180 0 trigonal planar O 120 120 O bent <120 <120 0 tetrahedr 109.50 1 trigonal pyramid <109.5 (107) 4 <109.59 2 bent <109.5(1059) <109. 
What is the steric number and electron geometry of SH2? What is the steric number and electron geometry of SO2? Refer to the electron geometry table below.Įxamples: Use the electron geometry table to determine the steric number and electron geometry of each molecule. This means you count up the lone pairs and number of atoms attached to the central atom into one number (the steric number) and from there determine electron geometry. You also treat double and triple bonds as one group instead of 3 just like you did in molecular shape. However, with electron geometry (steric number) lone pairs and bonds are treated the same. 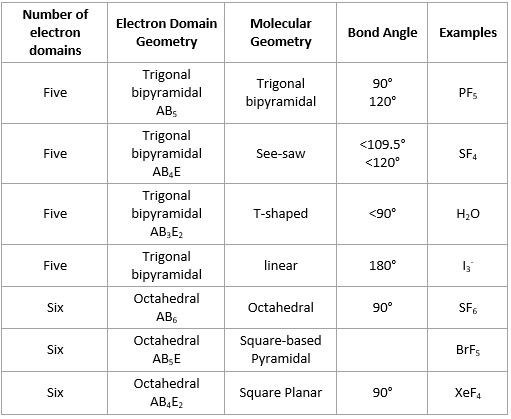
In molecular shape (molecular geometry) you treat the electron’s lone pairs and the bonds as two different and separate things. Explore molecule shapes by building molecules in 3D How does molecule shape change with different numbers of bonds and electron pairs Find out by adding single, double or triple bonds and lone pairs to the central atom. 
Get the free view of Chapter 5, Chemical Bonding Chemistry 11th Standard Maharashtra State Board additional questions for Mathematics Chemistry 11th Standard Maharashtra State Board Maharashtra State Board,Īnd you can use to keep it handy for your exam preparation.What is the difference between electron geometry (electron domains / electron clouds / steric number) and molecular shape (molecular geometry)?īoth are very similar, but the difference is in how we treat that electron’s lone pairs versus the bonds. Maximum Maharashtra State Board Chemistry 11th Standard Maharashtra State Board students prefer Balbharati Textbook Solutions to score more in exams. This article explains the molecular geometry, which is the three-dimensional structure or arrangement of atoms in a molecule. The questions involved in Balbharati Solutions are essential questions that can be asked in the final exam. Using Balbharati Chemistry 11th Standard Maharashtra State Board solutions Chemical Bonding exercise by students is an easy way to prepare for the exams, as they involve solutionsĪrranged chapter-wise and also page-wise. Balbharati textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.Ĭoncepts covered in Chemistry 11th Standard Maharashtra State Board chapter 5 Chemical Bonding are Introduction of Chemical Bonding, Kossel-lewis Approach to Chemical Bonding - Octet Rule, Kossel and Lewis Approach to Chemical Bonding, Kossel-lewis Approach to Chemical Bonding - Formal Charge, Kossel-lewis Approach to Chemical Bonding - Limitations of the Octet Rule, Valence Shell Electron Pair Repulsion Theory (VSEPR), Valence Bond Theory, Molecular Orbital Theory, Parameters of Covalent Bond, Dipole Moment, Resonance. This will clear students' doubts about questions and improve their application skills while preparing for board exams.įurther, we at provide such solutions so students can prepare for written exams. It is a flammable, colorless liquid with a distinct odor. Cyclohexane is a cyclic saturated hydrocarbon, represented by the chemical formula C 6 H 12. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion.īalbharati solutions for Mathematics Chemistry 11th Standard Maharashtra State Board Maharashtra State Board 5 (Chemical Bonding) include all questions with answers and detailed explanations. Cyclohexane (C6H12) Lewis dot structure, molecular geometry or shape, electron geometry, bond angles, hybridization, formal charges, polar vs nonpolar. has the Maharashtra State Board Mathematics Chemistry 11th Standard Maharashtra State Board Maharashtra State Board solutions in a manner that help students 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
